SciGen Teacher Dashboard
Unit M3
Close-up Look at Change
Concentrating on Pink Lemonade
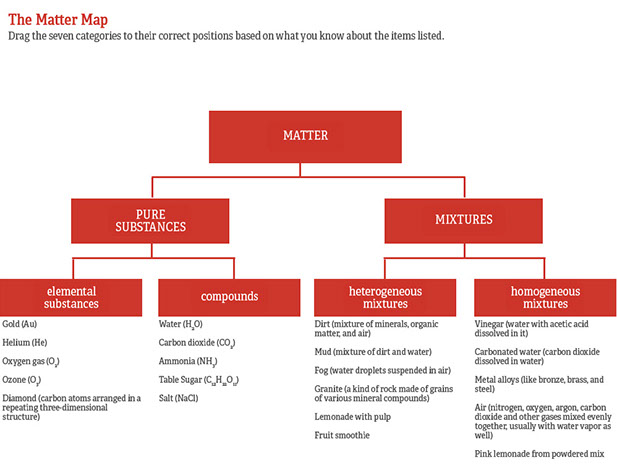
Categories of Chemicals and Mixtures
Condensation and Evaporation
Describing Physical and Chemical Change
The Three Little Chemists and the Big Bad Wolf
 Lesson: Categories of Chemicals and Mixtures
Lesson: Categories of Chemicals and Mixtures
Duration: Approximately 40 minutes
This activity introduces students to the classification of matter into pure substances and mixtures by using familiar examples. Students begin to consider which of the mixtures are homogeneous (same throughout) and which are heterogeneous (not evenly distributed). Within the "pure substances" category, students will note how some are elemental substance (made of just one element) and some are compounds (more than one kind of atom, but combined together in specific proportions).
As students become familiar with these categories, they will be able to use a tree diagram to map the examples.
LEARNING OBJECTIVE
Students will differentiate pure substances (chemicals) and mixtures.
Students will differentiate homogeneous mixtures (solutions) and heterogeneous mixtures.
Students will differentiate elemental substances and compounds.
Materials (one per student or group)
- electronic device (such as computer, laptop, tablet) to use the interactive elements in this activity
Teacher Tune-ups
Teaching Notes
ACTIVITY OVERVIEW
- Describe pure substances and mixtures (15 minutes)
- More examples (10 minutes)
- Matter map assessment (15 minutes)
Describe pure substances and mixtures (15 minutes)

This activity is about classification of matter into pure substances and mixtures. It provides background information for the interactive activity below.
Among other things, students learn that solutions are not always liquid: homogeneous mixtures can also take gaseous or solid forms. (In the case of a solid solution, like a metal alloy, the different chemicals typically become mixed in the liquid phase and later harden into a solid.)
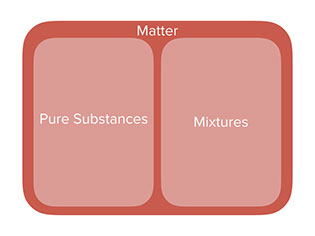
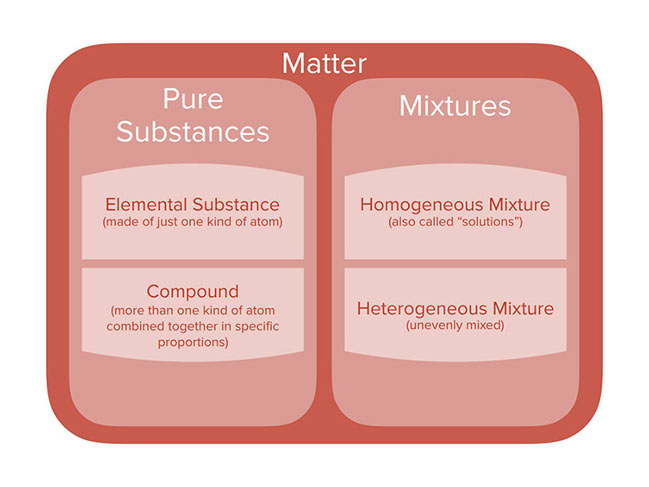
First show the diagram’s various stages that show how different categories of matter fit into one another. In this diagram, more specific categories are nested inside of more general ones.
:
Pretty much all the materials around us, and even in us, fit into one of two categories:
- Pure Substances (also called chemicals)
- Mixtures (combine multiple chemicals with chemcial reaction)
Within pure substances category there are:
- Elemental Substances (made of just one kind of atom)
- Compounds (combine multiple atoms in definite proportions)
Within the mixture category there are:
- Homogeneous Mixtures (also called "solutions")
- Heterogeneous Mixtures (unevenly mixed)
More examples (10 minutes)



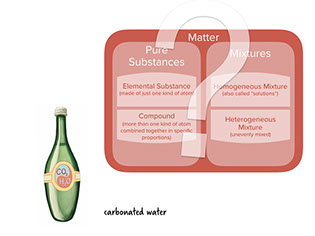
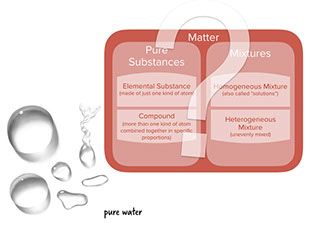

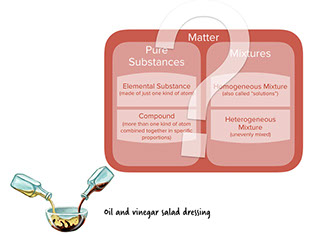
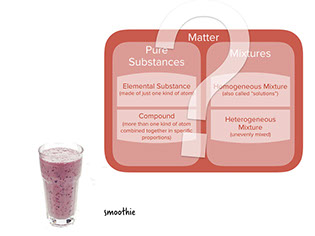

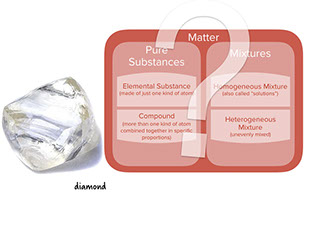
:
To practice classifying various familiar objects, we will play a Turn and Talk sorting game during which all teams will be shown an object. Then your team will discuss which of the categories corresponds to the object. Some of the answers will be surprising, so between each slide we’ll read a brief explanation about WHY the item fits into its category.
Teachers can choose whether or not to keep score.
:
- A gold bar is made of just one thing: the element gold.
- Bronze is a homogeneous mixture of copper and tin. It’s a mixture (not a compound) because the proportions of copper and tin aren’t exactly the same in all bronze. Mixtures of metals are called alloys.
- Steel is an alloy made by mixing a little carbon, and sometimes other elements, into iron. The term alloy usually means a mixture of metal, but adding carbon to iron is also considered an alloy, even though carbon is not a metal.
- Sometimes gases can be dissolved in liquids. For example, carbonated drinks have carbon dioxide dissolved in water, making a homogeneous mixture.
- Water is a chemical substance that contains only molecules made of two hydrogen atoms and one oxygen atom, so it's a compound.
- Table salt crystals are made of sodium and chlorine atoms in a one-to-one ratio. That is, there are always equal numbers of the two kinds of atoms. The scientific name for this salt is sodium chloride, which is a compound.
- Oil and vinegar don’t dissolve into one another, so they always remain unevenly mixed. It's a heterogeneous mixture.
- In a smoothie, you can see small pieces of chopped up fruit mixed into the liquid. It's a heterogeneous mixture.
- Concrete is made by mixing coarse material like broken stone, gravel, or sand into cement. It's a heterogeneous mixture.
- Diamond is made of just one element: carbon. Each carbon atom in diamond is connected to four other carbon atoms, in a crystal that extends on and on. There are other forms of pure carbon where the atoms are bonded differently, notably charcoal and graphite.
Matter map assessment (10 minutes)
BETA Version - Please send comments and corrections to info@serpinstitute.org