SciGen Teacher Dashboard
Unit M2
Determining Density
Archimedes & the Case of the Missing Gold
The Basics of Density
Heating Up and Spreading Out
Is Putty or Clay More Dense?
 Conversation: The Basics of Density
Conversation: The Basics of Density
Duration: Approximately 40 minutes
This activity centers around a fictional situation in which one person is asking another a question about wood and gold. The story moves through a series of typical challenges children have in thinking about density as a property. Ultimately students are given questions to work through that involve calculating the densities of various samples.
LEARNING OBJECTIVE
Students will connect fundamental density concepts with the common formula used to determine density: density = mass/volume.
Students will use the volume calculations in order to compare single cubic units appropriately.
Teacher Tune-ups
Teaching Notes
ACTIVITY OVERVIEW
- Introduce the density concept (20 minutes)
- The handy density formula (10 minutes)
- Worksheet: density fundamentals (10 minutes)
Introduce the density concept (20 minutes)
Facilitate a light-hearted discussion with many pauses for students to turn and talk as you display the 12 slides in this section.
:
- How do you interpret this cartoon?
- How do you interpret this cartoon?
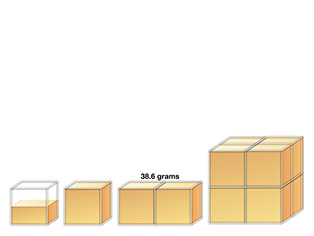
- Here we have four "samples" of gold. How much is each one?
- Does this help?
- Does this help more?
- If we know the mass of one of the samples, can we determine the mass of them all? How?
- What's the volume of this?
- Does this help?
- Predict: Which is heavier?
- Does this help you predict?
- How about this? (click on images to start animation)
- How do you interpret this cartoon?
Define density: Mass per unit volume (cubic centimeters is a frequent unit of volume used)
The handy density formula (10 minutes)
:
Are all objects shaped such that you can easily determine their volumes by looking (as was the case with the slides we just looked at)?
:
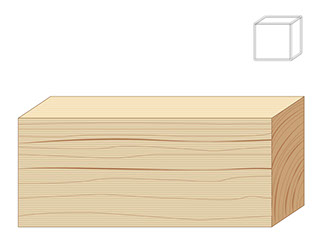
Of course not. There are many great ways to determine the volume of an object. For example, consider this rectangular prism.
Show the slide. :
It's difficult to tell the volume by looking because of the decimal values. But as many of you know from math class,
volume = length x width x height.
Once you know the volume of an object, it's not difficult to determine its density. You simply weigh the object and then do the math to figure out what a single cubic centimeter would weigh. That's the way we often talk about density in science: the mass of 1 cubic centimeter of a substance.
Allow students to think that explanation through and discuss with one another.
Lead the class to the formula for density:
density = mass/volume
:
The glass brick has a volume of 85.68 cubic centimeters. It has a mass of 214.2 grams.
214.2/65.68 = 2.5
Therefore the density of this glass is 2.5 grams per cubic centimeter.
Caution: So far the examples have been only solid, regularly shaped items. This unit includes more on oddly shaped solids–plus liquids and gases–in other activities.
Worksheet: density fundamentals (10 minutes)
This worksheet applies what the students have learned from the previous two sections, and also introduces water and ice into the density conversation.
:
- All of the answers to the four questions are 19.3 grams per cubic centimeter
- The density of gold is 19.3 g/cm3.
- The density of pine is 0.4 g/cm3.
BETA Version - Please send comments and corrections to info@serpinstitute.org